Actovegin — Tablets & Injections
Actovegin is a clinically used metabolic agent based on deproteinized hemoderivative of calf blood, obtained through ultrafiltration. It supports oxygen utilisation at the cellular level and is widely prescribed in neurological rehabilitation, peripheral vascular disorders, and diabetic polyneuropathy management.
Authentic product
Fast international shipping
Secure payment
Buy Actovegin — All Available Forms
All products are sourced directly from licensed pharmaceutical manufacturers. Choose your preferred dosage form below.

Actovegin 200 mg Tablets
Oral metabolic support formulation for long-term therapy protocols in peripheral circulation and neurological recovery.

Actovegin 400 mg Injections
High-concentration injectable solution used in intensive supportive treatment and acute vascular therapy protocols.
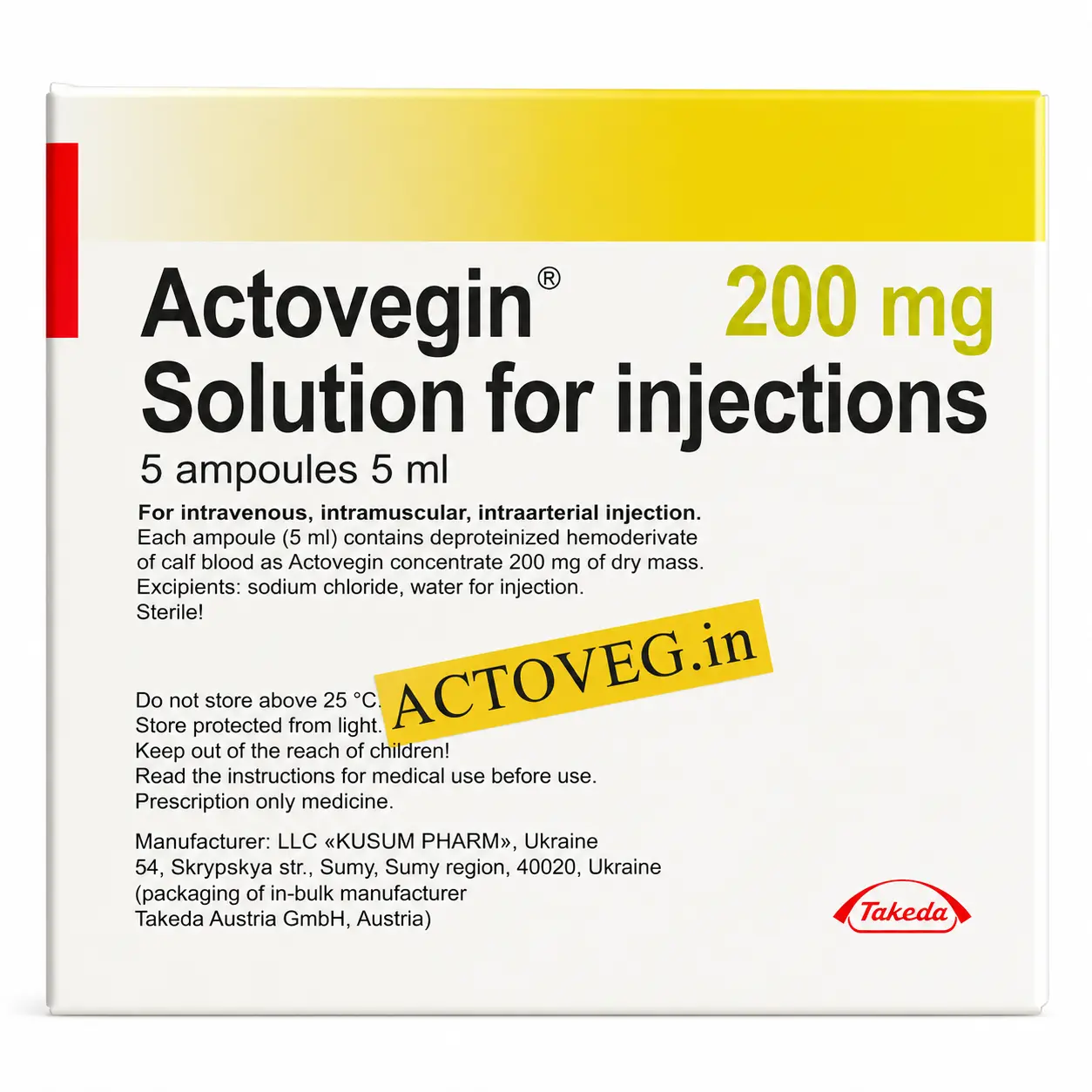
Actovegin 200 mg Injections
Standard injectable dosage commonly prescribed in supportive metabolic and peripheral vascular therapy.
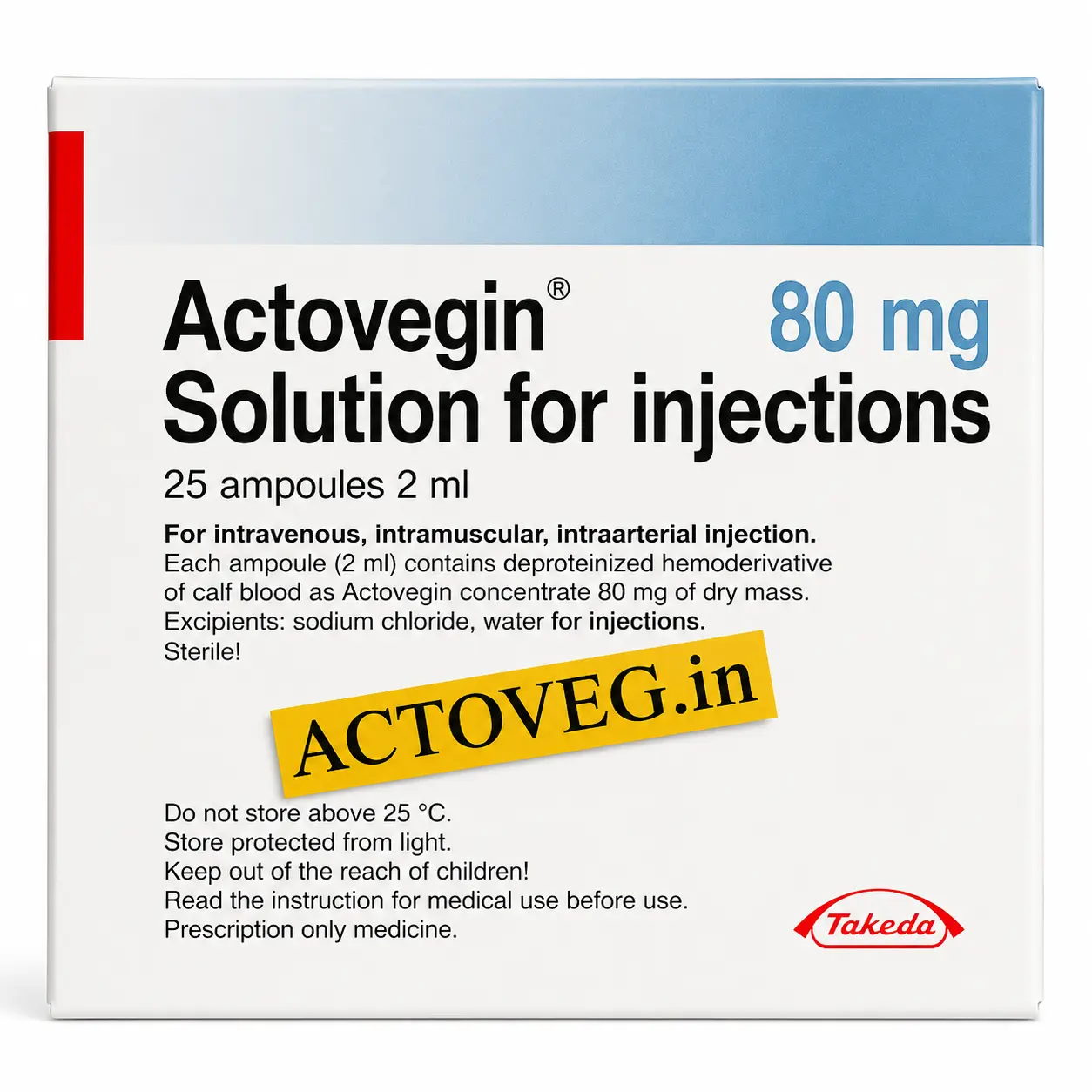
Actovegin 80 mg Injections
Compact ampoule format suitable for individualized low-dose supportive administration and maintenance protocols.
What Is Actovegin? Medical Overview
Actovegin is a biological medication derived from deproteinized hemoderivative of calf blood, manufactured
via advanced ultrafiltration technology. The active fraction consists of low-molecular-weight organic compounds
including amino acids, oligopeptides, nucleosides, and essential electrolytes that play a key role in
cellular energy metabolism and oxygen utilisation.
Unlike many synthetic drugs, Actovegin acts at the mitochondrial level — it enhances glucose uptake and
oxygen utilisation in tissues with impaired circulation or under metabolic stress. This mechanism makes
it particularly valuable in conditions where tissue hypoxia plays a central role.
Actovegin is registered as a prescription medication in numerous countries across Europe, Asia, and Latin
America, and is actively used in clinical neurology, angiology, and endocrinology departments.
Available Forms
- Actovegin 200 mg tablets (50 pcs)
- Actovegin 80 mg injections (25 ampoules)
- Actovegin 200 mg injections (5 ampoules)
- Actovegin 400 mg injections (5 ampoules)
- Topical gel, cream, and ointment
Clinical Applications
- Peripheral arterial circulation disorders
- Diabetic polyneuropathy
- Neurological and cognitive rehabilitation
- Metabolic recovery after stroke or TBI
- Tissue oxygen utilisation support
Frequently Asked Questions About Actovegin
What is Actovegin used for?
vascular disease, diabetic polyneuropathy, neurological rehabilitation after stroke or traumatic brain
injury, and general metabolic recovery. It is prescribed both as standalone therapy and as part of
comprehensive treatment protocols.
What forms of Actovegin are available?
injectable ampoule solutions in 80 mg/2 ml, 200 mg/5 ml, and 400 mg/10 ml strengths, as well as
topical formulations including gel, cream, and ointment for wound healing support.
Can Actovegin injections be self-administered at home?
sterile technique and appropriate equipment. Intramuscular or intravenous injection requires professional
training. Always consult your healthcare provider before starting any injectable therapy.
How should Actovegin be stored?
excessive moisture, and heat sources. Keep out of reach of children. Opened ampoules must not be
reused — discard any unused solution immediately after administration.
Which Actovegin dosage strength should I choose?
physician. Lower concentrations (80 mg) are used for maintenance or individualised dosing. The 200 mg
injectable form is the most commonly prescribed. High-dose 400 mg ampoules are reserved for intensive
intravenous therapy in clinical settings.
Is Actovegin the same as Solcoseryl?
but are manufactured by different companies (Takeda and Meda Pharma respectively) and may differ in
exact composition, excipients, and registered indications depending on the country.
