$47.00
Actovegin is a medication that is used for the following medical purposes:
- Improving blood circulation and oxygen supply to tissues
- Treatment of acute and chronic circulatory disorders
- Recovery from tissue damage or trauma
- Adjunctive therapy in sports medicine
Discount per Quantity
| Quantity | 10 - 19 | 20 - 39 | 40 - 69 |
|---|---|---|---|
| Discount | $2.00 | $3.00 | $4.00 |
| Price | $45.00 | $44.00 | $43.00 |
Description
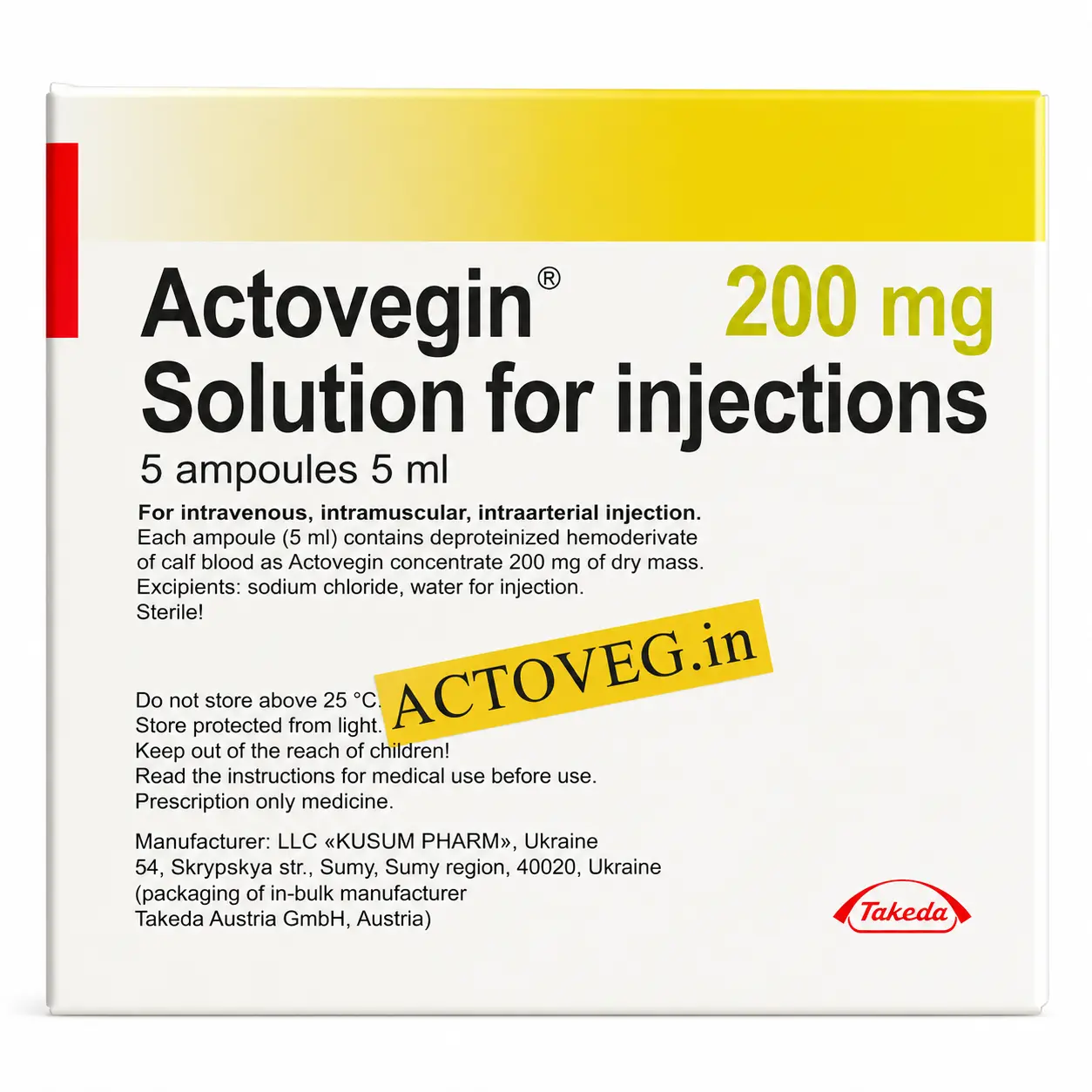
ACTOVEGIN® 200 mg (40 mg/mL), 5 mL – Solution for Injection (5 ampoules)
INN: Actovegin (deproteinized calf blood hemoderivative)
Therapeutic Category: Antihypoxant, metabolic and microcirculatory stimulator
Form: Sterile solution for injection, 40 mg/mL, 5 mL ampoules
Packing: 5 ampoules in tamper-evident packaging with patient leaflet
Manufacturer: Takeda Pharmaceuticals International AG (Japan/Austria)
Composition
- Active: Deproteinized calf blood hemoderivative – 40 mg/mL (dry matter)
- Excipients: Water for injection; sodium chloride approximately 26.8 mg per 5 mL
Description
Sterile, clear to slightly yellow solution in glass ampoules with break points. No visible particles. pH ~7.0. Osmolarity near physiological levels.
Pharmacodynamics & Mechanism
Actovegin is ultrafiltered calf hemoderivative with low molecular weight fractions (<5000 Da). It exerts metabolic, neuroprotective, and microcirculatory effects. Enhances cellular oxygen and glucose uptake, reduces lactate formation, stimulates ATP production. Modulates NF‑κB and inhibits PARP and caspase‑3 pathways, protecting neural and endothelial cells. Improves capillary perfusion by reducing arteriolar tone and enhancing erythrocyte flexibility.
Clinical onset within ~30 minutes; peak effects at ~3 hours post-injection.
Indications
- Ischemic and metabolic disorders of the brain (e.g., stroke, cerebral insufficiency, dementia, TBI)
- Peripheral vascular disorders (angiopathy, venous insufficiency, trophic ulcers)
- Diabetic peripheral neuropathy (DPN) – symptomatic relief and improved microcirculation
- Radiation-induced tissue damage (skin, mucous membranes)
- Wound care post-burns or skin grafts
Dosage & Administration
Administer under sterile conditions. Test injection for hypersensitivity recommended.
- General: 10–20 mL IV/IA daily; maintenance 5 mL IV slowly or IM daily or 3–4 times weekly
- Infusion: Dilute 10–50 mL in 200–300 mL isotonic saline or 5% dextrose; infusion rate ~2 mL/min; use ≥15 μm filter
- Vascular brain disorders: 250–500 mL/day IV for 2 weeks, then 250 mL IV 2–3×/week for ≥4 weeks
- DPN regimen: 2,000 mg/day IV for 3 weeks, followed by tablets 3×3/day (~1,800 mg/day) for 4–5 months
- Ulcers/Burns: 10 mL IV or 5 mL IM daily, combined with topical therapy
Immediately discard opened ampoule; do not store.
Pharmacokinetics
No classical pharmacokinetic measurements – Actovegin components are endogenously derived and rapidly cleared via metabolism.
Contraindications & Warnings
- Hypersensitivity to product or excipients
- Pulmonary edema, oliguria, anuria, fluid overload
- Uncompensated heart failure
Use with caution in electrolyte imbalances (hyperchloremia, hypernatremia). Monitor cardiovascular status during IV use.
Adverse Effects
Generally well tolerated. Rare skin reactions (rash, urticaria); very rare anaphylaxis. No toxicity reported at up to 30–40× doses.
Use During Pregnancy & Lactation
No controlled human data. Use only if benefits justify potential risk.
Use in Pediatric Patients
Not recommended due to lack of safety and efficacy data.
Effect on Driving & Machinery
No known detrimental effects.
Clinical Evidence
- ARTEMIDA trial (503 post-stroke patients): improved cognition at 6 months (ADAS‑Cog+ –6.8 vs –4.6; P=0.005); safety similar to placebo (PMID 28432265)
- 2022 systematic review (~3,800 patients): uncertain net benefit; possible increased stroke recurrence (PMID 35771887)
- Mechanistic review (2020 IJMS): antioxidative, anti-inflammatory, anti-apoptotic pathways (PMID 32365943)
Use in Sports and Athletic Performance
Actovegin® has drawn significant interest in the field of sports medicine due to its proposed effects on oxygen utilization, energy metabolism, and tissue regeneration. Although it is not classified as a performance-enhancing drug by WADA, it has been historically used in both professional and amateur sports contexts—particularly for recovery from injuries and adaptation to intense physical loads.
Anecdotal and Practical Use Among Athletes
On various sports forums, Actovegin® is referred to as a substance used for “oxygen loading” or as a recovery aid. Athletes in endurance disciplines such as cycling and long-distance running have reported subjective benefits, including increased stamina and faster recovery times. A discussion on LetsRun.com details accounts of athletes combining Actovegin with other agents prior to competition to reduce fatigue and improve performance, although these reports remain anecdotal.
Clinical and Scientific Data
- A 2024 double-blind, placebo-controlled study published in Nutrients examined cardiopulmonary responses in trained athletes following 7-day oral Actovegin administration. While trends suggested possible physiological changes (e.g., oxygen pulse, ventilatory thresholds), no statistically significant improvements were observed over placebo. PubMed 39408298
- A clinical trial involving professional footballers reported that intramuscular administration of Actovegin shortened return-to-play after muscle injuries by ~8 days compared to physiotherapy alone (p = 0.033), without adverse events. Thieme Connect
- Preclinical studies in diabetic mice and muscle cultures suggest Actovegin enhances glucose uptake, mitochondrial respiration, and cell survival, potentially benefiting muscle recovery. PubMed 37913525
Expert Commentary and Reviews
A 2024 review published in Experimental and Applied Biomedical Research concludes that while robust performance-enhancing evidence is lacking, Actovegin has clinical promise in localized injury management and rehabilitation protocols. It has been used in sports medicine for over 20 years with a favorable safety profile when applied intramuscularly. Sciendo Article
Another 2020 review in the International Journal of Molecular Sciences emphasized Actovegin’s pleiotropic effects—anti-inflammatory, anti-apoptotic, and antioxidative—especially relevant in ischemic and hypoxic tissue states seen in high-intensity training. PubMed 31855916
WADA Compliance and Anti-Doping Regulations
- Actovegin is not a banned substance on its own; however, intravenous infusions exceeding 100 ml within 12 hours are prohibited by the World Anti-Doping Agency under Code M2. WADA Website
- Intramuscular administration, which is the most common therapeutic route in sports injury recovery, remains permitted but must be medically justified.
- Athletes using Actovegin should maintain full medical documentation and consult with team physicians to avoid doping violations.
Summary for Athletes and Trainers
- Actovegin may contribute to faster muscle regeneration and tissue healing following injury.
- Evidence of improved endurance performance is limited and inconsistent across studies.
- Not prohibited by WADA, but its use must follow infusion volume rules and medical best practices.
- Recommended primarily in sports injury recovery protocols under professional medical supervision.
- Preclinical models: enhanced mitochondria and glucose uptake without toxicity (PMID 37913525)
Patient Advice
- Complete treatment even if symptoms subside
- Monitor blood pressure and hydration with parenteral use
- Report allergies immediately
- Pregnant or breastfeeding individuals should consult physician
- Ensure proper disposal of opened ampoules
Storage & Shelf Life
Store at ≤ 25 °C in original packaging, away from light and moisture. Shelf life: 36 months. Keep out of reach of children.